BMC Medical Education volume 24, Article number: 1202 (2024)
Abstract
As clinical trials for psychedelics move into phase III in the USA, Europe must address its lag in integrating professional education around psychedelic-assisted therapy (PAT) and supporting psychedelic drug research. This paper evaluates the necessary frameworks for implementing PAT in Germany, emphasizing the nation’s potential leadership role within the European Union. With Australia having already approved MDMA and psilocybin for mental health indications, the Ukrainian government exploring MDMA treatment for war-related PTSD, and initial clinical trials involving MDMA and LSD with patients in Switzerland which restarted the restricted medical use of these substances around 2014, the medical authorization of psychedelics in these countries establishes precedent showcasing both the promise and challenges of researching and implementing PAT in nations where the substances were formally scheduled as illicit substances. Key challenges include establishing rigorous standards for practitioner training, accessibility, and defining regulatory oversight. This paper focuses on the development of robust infrastructure in Germany, which will support the roll out of PAT, and details ethical considerations, training protocols, and governmental roles in the formulation of treatment frameworks. This approach aims not only to guide Germany in adopting PAT but also to influence broader European policy, ensuring that patients receive ethically sound and proficient care. The findings suggest pathways for Europe to reclaim its historical lead in psychiatric and therapeutic innovation.
Introduction
Psychedelic-assisted therapy (PAT) is a modality of mental health treatment that merges psychotherapeutic interventions with psychedelic states, often facilitated by substances such as lysergic acid diethylamide (LSD), psilocybin, and 3,4-Methylenedioxy-methamphetamine (MDMA). The latter two being in phase III trials. Whereas MDMA is considered an entactogen that enhances self-awareness and emotional connectivity, psilocybin is a naturally occurring psychedelic compound found in certain mushrooms. Recent research suggests that these and other psychedelics, all small molecules, most with benzene or phenyl rings, uniquely work by reopening a “critical period” in the brain, allowing for new learning within social contexts in a process involving changes in brain plasticity and oxytocin signaling [1]. Despite their classification as a Schedule I drugs under the Controlled Substances Act by the Drug Enforcement Agency (DEA) in the United States, their therapeutic potential has been increasingly recognized, with demands from the public to make them available for those with treatment resistant conditions. These among other developments resulted in MDMA being granted Breakthrough Therapy designation by the U.S. Food and Drug Administration (FDA) for the treatment of treatment-resistant PTSD (TR-PTSD) in phase II, (phase III studies were for ‘moderate to severe’ PTSD). Psilocybin (COMP360) and a deuterated psilocybin analogue (CYB003) were granted FDA Breakthrough Therapy designation for treatment resistant depression (TRD) [2,3,4].
Methodologically rigorous clinical research suggests that PAT may offer substantial long-term alleviation of symptoms in patients suffering from psychopathologies such as PTSD, TRD, major depressive disorder (MDD), end of life anxiety, obsessive–compulsive disorder (OCD), substance use disorders (SUD), psychotic conditions, and more [5, 6]. Notably, a single session of substance-assisted therapy has been reported to lead to significant symptom reduction, with some patients achieving remission that can persist for at least twelve months [7]. This enduring effect underscores the potential of psychedelic-assisted therapy in providing therapeutic outcomes that are significantly superior to antidepressant medications such as SSRIs, which often have poor efficacy rates and unwanted side-effects such as agitation, weight gain, sexual performance difficulties, gastrointestinal issues, and other symptoms [1, 8].
In February 2024, Lykos (formerly MAPS PBC) submitted a new drug application (NDA) to the FDA for MDMA (Midomafetamine) capsules in combination with psychotherapy for the treatment of PTSD [9]. Following the submission, a citizen petition was filed, raising concerns about the integrity of the clinical trials. This prompted the FDA to convene an advisory board meeting in June 2024 to review the claims. The petition raised several issues, including alleged bias in the trial design, inadequate sample diversity, insufficient double-blinding, underreporting of adverse events such as sexual misconduct, and confusion regarding the integration of psychotherapy within the study design [10]. Despite the principal view that MDMA trials are sound, and even the FDA’s participation in the creation and oversight of the studies these concerns influenced the FDA’s review process. Subsequently, in August 2024, the FDA issued a final decision rejecting the NDA under the Prescription Drug User Fee Act (PDUFA). It is important to note however that the FDA encouraged ongoing MDMA research and asked for another Phase III trial.
This decision led to additional actions, including the Journal of Psychopharmacology retracting three articles related to the MDMA clinical trials conducted by the MAPS research team and the initiation of an FDA investigation. Lykos has since filed for a reevaluation of their NDA. If approved, MDMA would become the first psychedelic-assisted therapy officially recognized in the United States.
As unexpected as the August 2024 rejection of the application by Lykos for approval of MDMA was for some, the hope based on the phase II studies, remains that either MDMA or psilocybin will within the next two to three years receive a positive evaluation from the FDA although the exact timing remains unknown. Unlike the MDMA trials, where the FDA was initially satisfied with the blinding process prior to the advisory board meeting, Compass’ psilocybin trials were designed to minimize the unblinding caused by psychotropic effects, following the FDA’s advice to their satisfaction until this day.
Lykos and Compass have been the two major companies driving development, and although Lykos has yet to submit a new phase III proposal, Compass continues with their phase III clinical trial. In general enthusiasm in the field for further research continues as there remains a need for novel treatments, and despite the uncertainty, the FDA seems generally favorable toward psychedelic medicine [10].
This paper outlines the current and required infrastructure for the successful integration of PAT, including rescheduling of psychedelic drugs beyond ketamine, accessibility, reimbursement strategies, accreditation of practitioners, ethical considerations and educational requirements. The role of the German government and affiliated agencies is pivotal in shaping this framework, ensuring that the setup not only complies with regulatory standards but also supports the ethical deployment of these therapies.
Moreover, with the European Medicines Agency (EMA) currently deliberating on the integration of psychedelics within the European framework, Germany has a unique opportunity to lead by example, showcasing a meticulous approach to the adoption of psychedelic-assisted therapies and must therefore also prepare to accommodate these innovative treatments [11]. This could serve as a model for other European nations, promoting a harmonized approach to these promising treatments across the continent.
The first half of this paper covers the regulatory environment in Germany, as it is impossible to understand the steps required to make PAT a reality without some in-depth understanding of the country’s unique health care system. The second half of this paper covers the German provision of outpatient mental healthcare and how and where PAT would fit and critically, proposes a training scheme for the education of PAT facilitators.
Historical roots of psychedelics in Germany
Psychedelic research in Germany harkens back to the 1910s and 1920s when the atypical psychedelic MDMA was first synthesized, and pioneering research was being conducted on the properties of mescaline. During this early period, Beringer and colleagues [12] saw in mescaline and similar substances, an opportunity to explore the phenomenology of psychopathology, creating what they described as ‘model psychoses.’ Although problematic in many ways, this stream of research opened up a new dimension of empathy and understanding into the experience of individuals with chronic psychosis [13]. In fact, the research carried out at the University of Heidelberg, culminating in Beringer’s habilitation thesis “Der Meskalinrausch” from 1927, can be considered the first major work in the field of psychedelic psychopharmacology in the West [12]. Another noteworthy event in the history of psychedelic drugs in German-speaking Europe is Albert Hoffman’s accidental discovery of the properties of lysergic acid diethylamide (LSD) on April 19, 1943, which accelerated interest in psychedelic compounds throughout the Western world [14]. In particular, this landmark event led to the widespread experimental use of psychedelics for a diverse range of psychiatric conditions across Europe and North America.
This period of research during the 1950s and 1960s, though short-lived, would later become known as the first wave of psychedelic research [14, 15]. During this brief moment in history, Betty Eisner, a German-educated American, first described the implementation of low-dose LSD in combination with psychotherapy, making a major contribution in the field which still today remains underrecognized [16]. Margot Cutner, a German psychoanalyst who was leading psychedelic research in England after fleeing from the Nazis, provided some of the first insights on the relevance of the role of the facilitator in psychedelic-assisted therapy (PAT) and the now well-known notion of ‘set and setting’ [16]. Following this, Hanscarl Leuner coined the term “psycholytic therapy” at the University of Göttingen underscoring the drug’s therapeutic potential in a sub-threshold dose range [17]. Despite Leuner and colleagues’ extensive research on LSD being among the most comprehensive bodies of work on the topic to date, it has been largely neglected until recently due to never being published in English [18].
A surprising turn of events occurred when in 1961, the United States passed the seemingly politically motivated US Controlled Substances Act, which resulted in an immediate and indefinite suspension of psychedelic research throughout the U.S. Europe was quick to follow suit, and psychedelics became labeled as potentially dangerous and addictive with no accepted medical use [19]. Subsequently, despite early breakthroughs and extensive research, these restrictions ushered in a prohibition era that would last decades, hampering progress and limiting the exploration of psychedelic compounds throughout the Western world. Germany was no exception, and psychedelic treatments now being championed for their therapeutic potential were outlawed.
Economic burden of treating PTSD and depression in Germany
The economic and human costs of PTSD and depression in Germany highlight an urgent need for more effective interventions [20]. Trauma-related healthcare costs range from 524.5 million to 3.3 billion euros annually [21], while depression adds another 1 to 5.2 billion euros [22, 23]. Current pharmaceutical treatments, such as serotonin-reuptake inhibitors (SSRI), offer limited efficacy and fail to fully address the needs of individuals with PTSD, depression, or their comorbidities [24].
A recent study of German insurance claims highlighted both the direct and indirect costs of PTSD (ICD-10-GM F43.1) [20, 21]. PTSD patients typically suffer for about 6 years, with a 50–100% likelihood of comorbid conditions such as major depressive disorder (MDD), panic disorder, and substance use disorder (SUD). Per-patient costs were 43,000 EUR, three times higher than for those without PTSD, driven by increased healthcare utilization, impaired work capacity and reduced quality of life. PTSD also accounts for approximately 200,000 Disability-Adjusted Life Years (DALYs) annually in Germany, a metric that reflects both premature mortality and years lived with disability, quantifying the overall burden of disease [25].
Similarly, depression carries significant economic burdens with indirect costs from labor absenteeism, social benefits, and prevention measures estimated at 10 to 16 billion euros annually, surpassing direct healthcare costs [26, 27]. Depression accounts for approximately 470,000 DALYs in Germany [28], while globally, PTSD contributes an additional 3 million DALYs, underscoring its substantial public health impact.
In short, PTSD and depression remain conditions with a high unmet need. SSRIs, first introduced in 1988 (fluoxetine), are still the primary pharmaceutical treatment for many psychological disorders, despite their limited efficacy and adverse side effects, including symptom exacerbation and suicidal thoughts [29].
Regulatory landscape
The European Medicines Agency (EMA) grants marketing authorization for new medicines across the EU. The sponsor of the medication submits an application for approval to the EMA following phase III trials, and after EMA approval, marketing authorization is granted, which allows the medication to be sold in all European Union member states. Sponsors then must decide which member states they wish to enter, as, even if the Sponsor has marketing authorization, each EU state has its own rules about how health insurers will be reimbursed for new medications. European member states furthermore have country specific processes and infrastructure around the provision of therapeutic services which are an essential part of PAT.
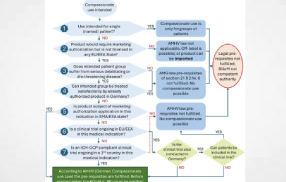
In Germany, the Bundesinstitut für Arzneimittel und Medizinprodukte (BfArM) oversees both clinical trial approval (pre- and post-EMA approval) and the documentation as well as considerations related to safety, efficacy, and quality, and specific labeling requirements tailored to the German context (Fig. 1).
Read full paper